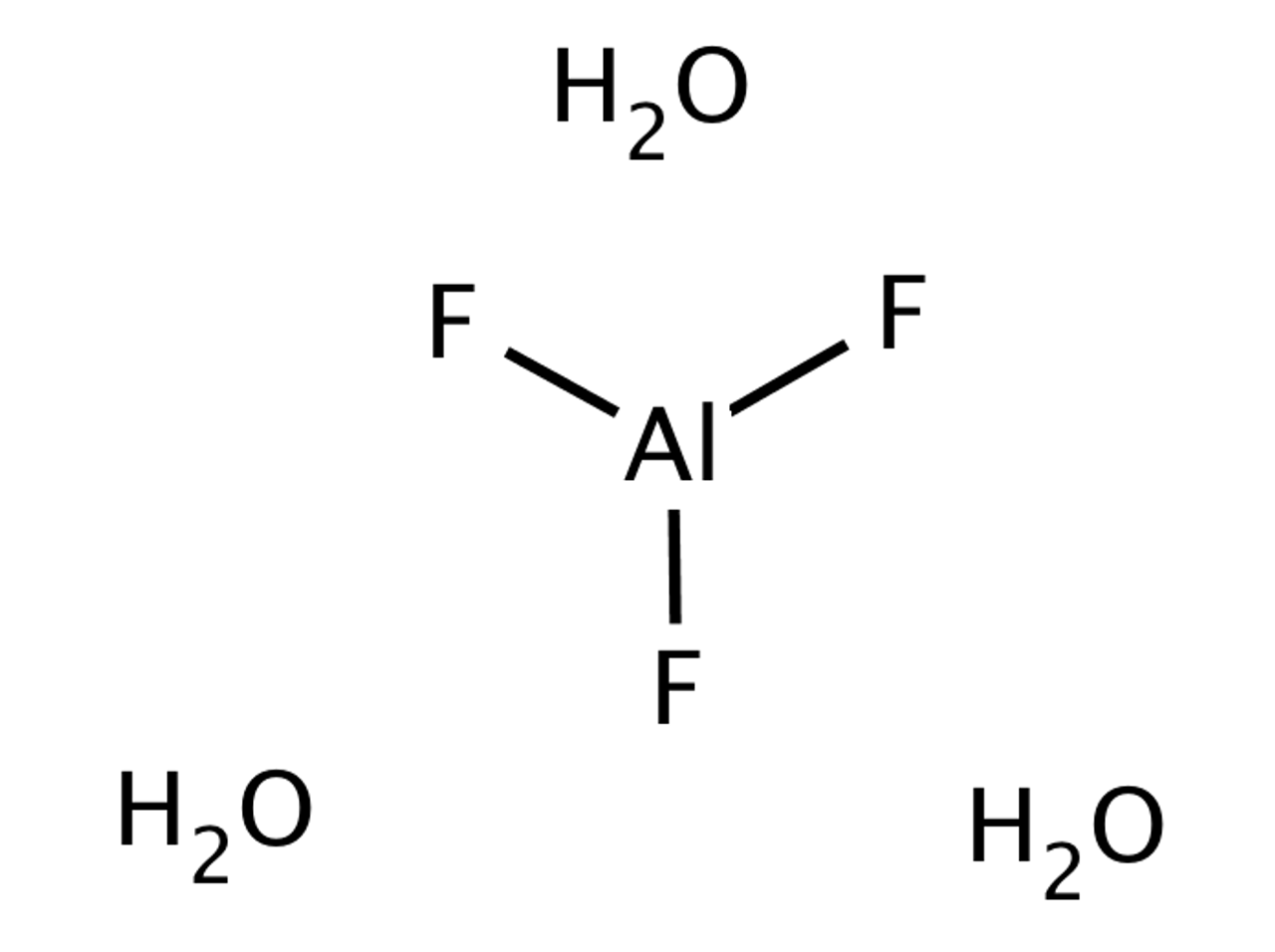
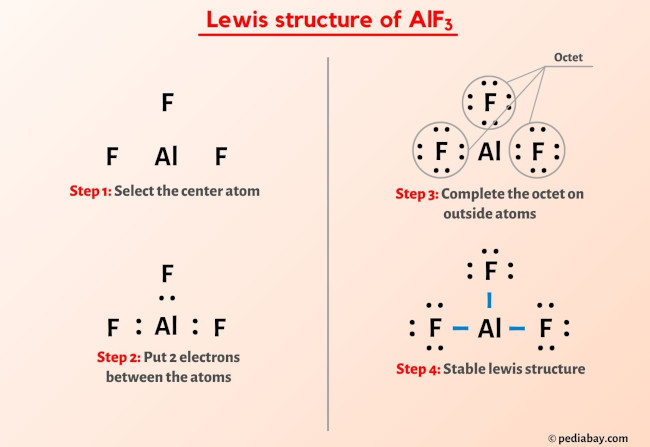
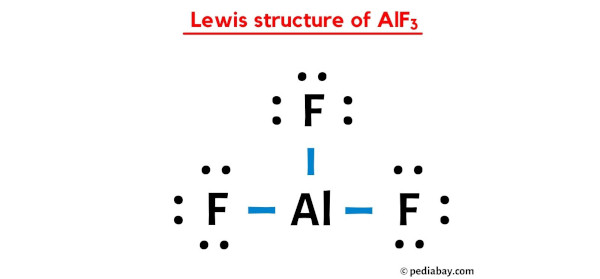
Possible Formation of H3O+ Cations Due to Aluminum Fluoride Interactions with Water. | Semantic Scholar

Possible Formation of H3O+ Cations Due to Aluminum Fluoride Interactions with Water. | Semantic Scholar

The molecular graphs of the AlF3-C2H2 (the top left), AlBr3-C2H4 (the... | Download Scientific Diagram

Possible Formation of H3O+ Cations Due to Aluminum Fluoride Interactions with Water. | Semantic Scholar

Atomic Layer Deposition of AlF3 Using Trimethylaluminum and Hydrogen Fluoride | The Journal of Physical Chemistry C
Inhibition of AlF3·3H2O Impurity Formation in Ti3C2Tx MXene Synthesis under a Unique CoFx/HCl Etching Environment

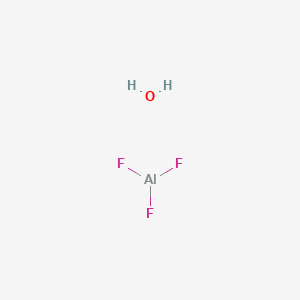
Hydrate de fluorure d'aluminium, Puratronic , 99,99 % (base métallique), Thermo Scientific Chemicals | Fisher Scientific

The acid strength of the datively bound complexes involving AlF3 lone pair acceptor and various lone pair donors - ScienceDirect
![PDF] Competition between Al2O3 atomic layer etching and AlF3 atomic layer deposition using sequential exposures of trimethylaluminum and hydrogen fluoride. | Semantic Scholar PDF] Competition between Al2O3 atomic layer etching and AlF3 atomic layer deposition using sequential exposures of trimethylaluminum and hydrogen fluoride. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e2ac2e88133be5a7278abdd433a3dd75d426a246/2-Figure1-1.png)
PDF] Competition between Al2O3 atomic layer etching and AlF3 atomic layer deposition using sequential exposures of trimethylaluminum and hydrogen fluoride. | Semantic Scholar
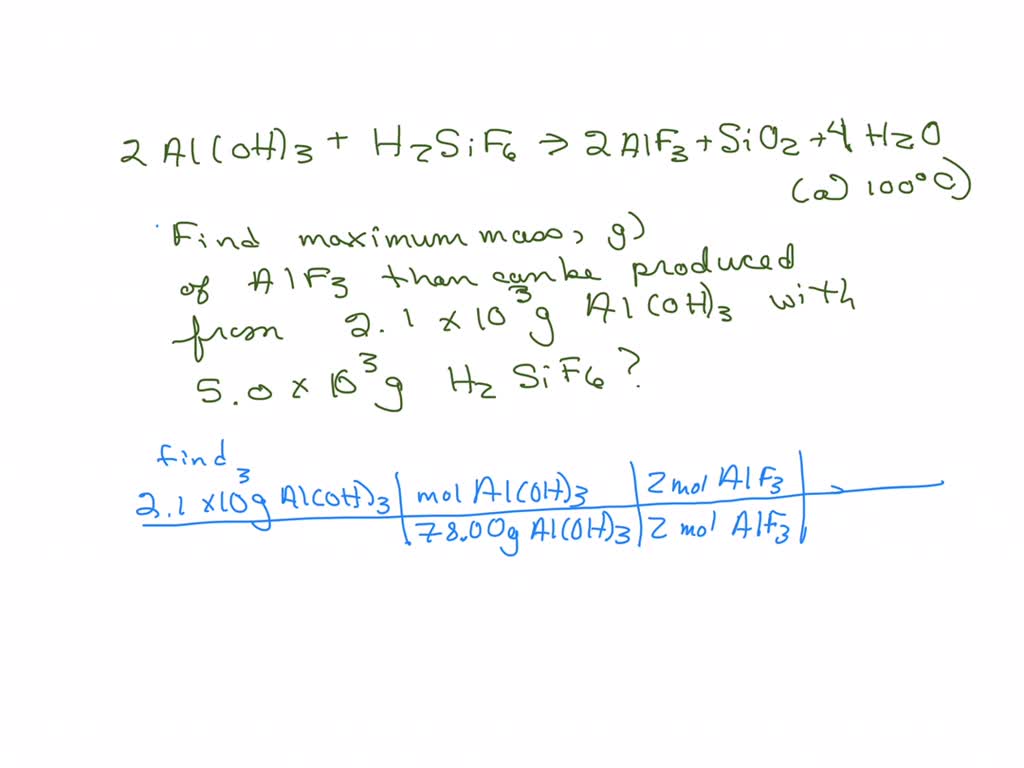
SOLVED: The equation for one process for making aluminum fluoride follows. What is the maximum mass, in grams, of aluminum fluoride, AlF3, that can be produced from the complete reaction of 2.1