![Ca2[Ti(HPO4)2(PO4)]·H2O, Ca[Ti2(H2O)(HPO3)4]·H2O, and Ti(H2PO2)3: Solid-State Oxidation via Proton-Coupled Electron Transfer | Inorganic Chemistry Ca2[Ti(HPO4)2(PO4)]·H2O, Ca[Ti2(H2O)(HPO3)4]·H2O, and Ti(H2PO2)3: Solid-State Oxidation via Proton-Coupled Electron Transfer | Inorganic Chemistry](https://pubs.acs.org/cms/10.1021/acs.inorgchem.1c02685/asset/images/acs.inorgchem.1c02685.social.jpeg_v03)
Ca2[Ti(HPO4)2(PO4)]·H2O, Ca[Ti2(H2O)(HPO3)4]·H2O, and Ti(H2PO2)3: Solid-State Oxidation via Proton-Coupled Electron Transfer | Inorganic Chemistry

HOW TO BALANCE H3PO4+Fe(OH)2=H2O+Fe3(PO4)2 | HOW TO BALANCE H3PO4+Fe(OH)2= H2O+Fe3(PO4)2 | By Chemistry 360 | Facebook

La vanmeersscheite U(UO2)3(PO4)2(OH)6 • 4 H2O et la méta-vanmeersscheite U(UO2)3(PO4)2(OH)6 • 2 H2O, nouveaux minéraux - Persée

SOLVED: 1. Balance the equation H3PO4 + Ca(OH)2 —> Ca3(PO4)2 + H2O and show the tally table. 2. Draw a colored particle diagram of your total balanced chemical equation. Show a key.
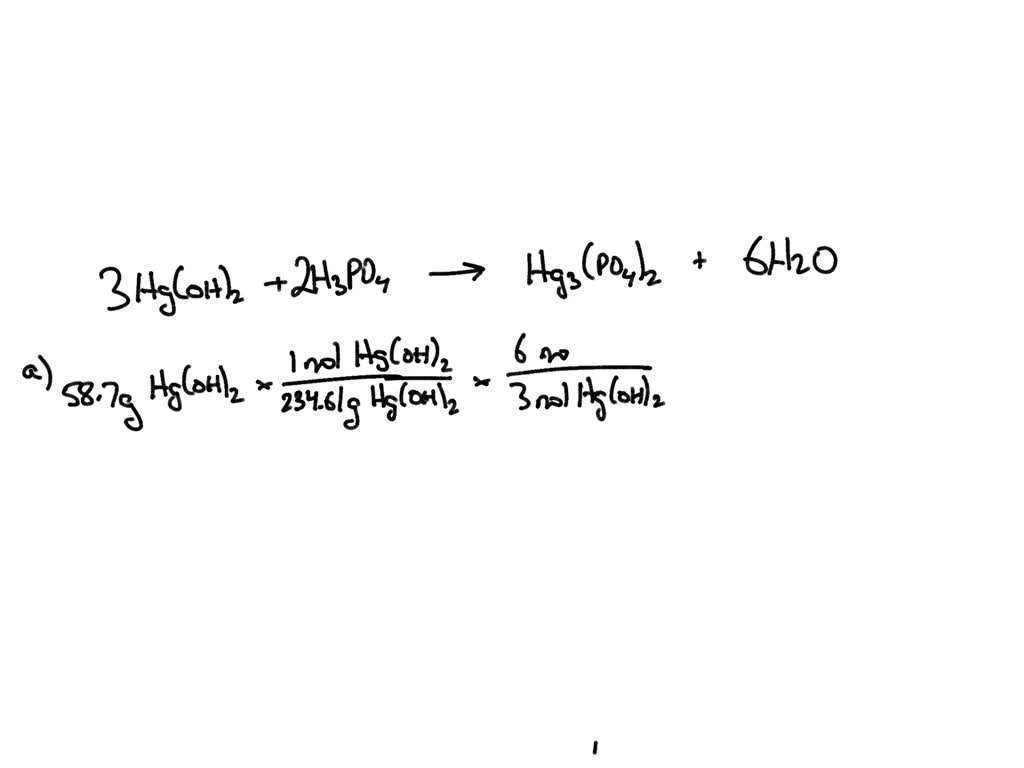
SOLVED: Use the reaction to solve the following problems. Hg(OH)2 + H3PO4→ Hg3(PO4)2 + H2O A) Given 58.7 grams of Hg(OH)2, how many moles of H2O will be produced? If you react

SOLVED: When the equation Ca(OH)2 + H3PO4 –> Ca3(PO4)2 + H2O is balanced, the coefficient for H2O is