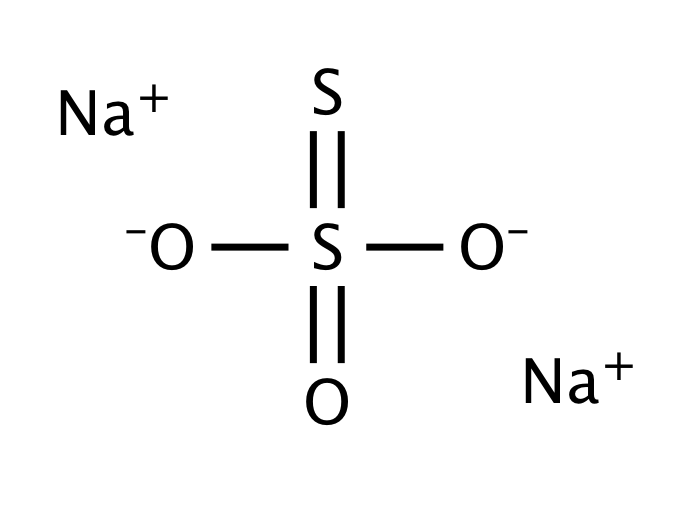48 Sodium thiosulphate (Na2S2O3 -5H20) reacts with iodine as follows: 28203- + 12 - S0+ 21 Calculate the equivalent mass of Na2S2O3.

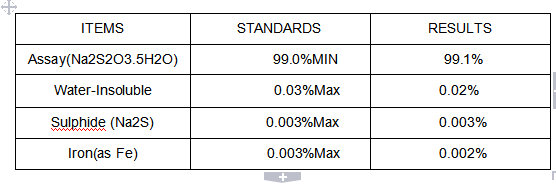
Na2S2O3 5H2O du thiosulfate de sodium Pharma Grade, cristaux incolores - Chine Le Sodium, de thiosulfate de sodium Thiosulfate de sodium

a) Sodium thiosulfate rod raw material (Na2S2O3 · 5H2O), a spherical... | Download Scientific Diagram

Uncaking cristaux incolores de qualité industrielle Na2S2O3 5H2O du thiosulfate de sodium - Chine Le thiosulfate de sodium anhydre, hyposulfite de sodium
What mass of Na2S2O3.5H2O is needed to make 500 cm^3 of 0.200 N solution for the reaction : - Sarthaks eConnect | Largest Online Education Community
![Amazon.com: Sodium Thiosulfate Pentahydrate [Na2S3O4.5H2O] 99% AR Grade Powder 8 Oz in a Bottle USA : Industrial & Scientific Amazon.com: Sodium Thiosulfate Pentahydrate [Na2S3O4.5H2O] 99% AR Grade Powder 8 Oz in a Bottle USA : Industrial & Scientific](https://m.media-amazon.com/images/I/81X8e-WucrL.jpg)
Amazon.com: Sodium Thiosulfate Pentahydrate [Na2S3O4.5H2O] 99% AR Grade Powder 8 Oz in a Bottle USA : Industrial & Scientific

Le thiosulfate de sodium, hyposulfite de sodium, Na2S2O3 5H2O - Chine Hyposulfite de sodium, de thiosulfate de sodium
The amount of Na2S2O3.5H2O required to completely reduce 100 mL of 0.25 N iodine solution, is (A) 6.20 g(B) 9.30 g(C) 3.10 g(D) 7.75 g

Le thiosulfate de sodium, hyposulfite de sodium, Na2S2O3 5H2O - Chine Hyposulfite de sodium, de thiosulfate de sodium
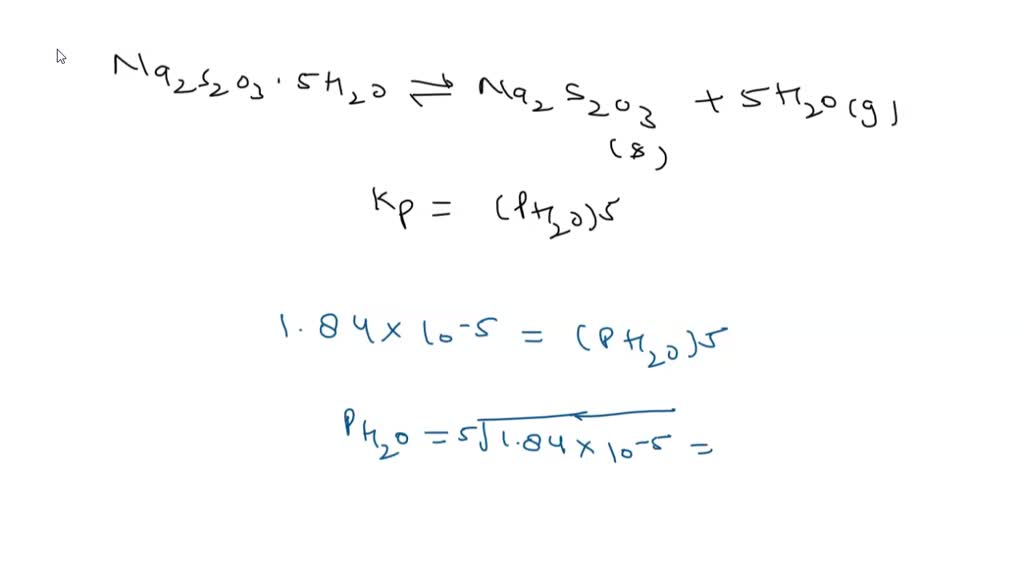
SOLVED: Sodium thiosulfate pentahydrate (FM 248.18 g ∙ mol-1) loses water when it is heated in an oven: Na2S2O3∙5H2O(s) ⇌ Na2S2O3(s) + 5H2O(g). ΔH° and ΔS° for this reaction at 25°C are