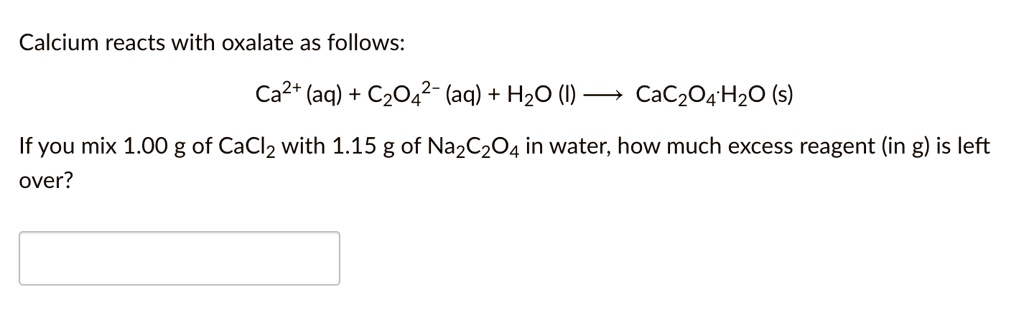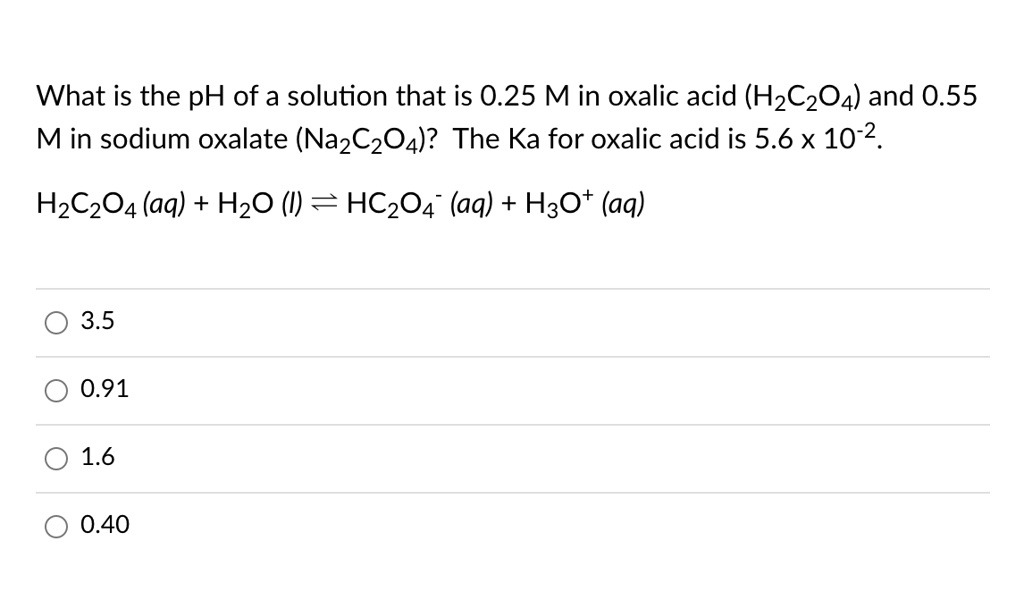
SOLVED: What is the pH of a solution that is 0.25 M in oxalic acid (H2C2O4) and 0.55 M in sodium oxalate (Na2C2O4)? The Ka for oxalic acid is 5.6 x 10^-2.

Plot of solubility against reciprocal temperature for sodium oxalate.... | Download Scientific Diagram

How to Write the Net Ionic Equation for H2C2O4 + NaOH = Na2C2O4 + H2O | How to Write the Net Ionic Equation for H2C2O4 + NaOH = Na2C2O4 + H2O Did
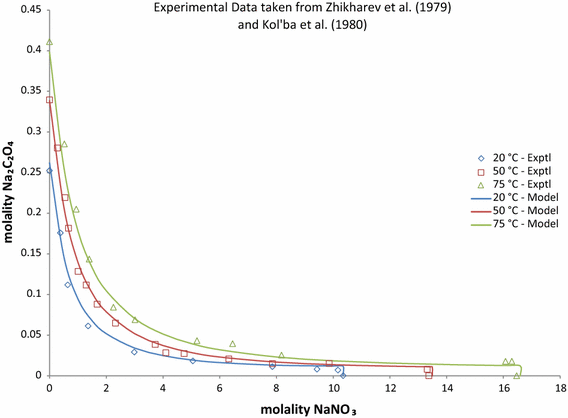
Pitzer Model Anion–Anion and Ternary Interaction Parameters for the Na2C2O4 –NaOH–H2O and Na2C2O4–NaNO3–H2O Systems | Journal of Solution Chemistry

SOLVED: A solution of sodium oxalate (Na2C2O4) in acidic solution is titrated with a solution of potassium permanganate (KMnO4) according to the following balanced chemical equation: 2KMnO4(aq) + 8H2SO4(aq) + 5Na2C2O4(aq) â†'

Solubility of Sodium Oxalate in Concentrated Electrolyte Solutions | Journal of Chemical & Engineering Data

SOLVED: A solution of sodium oxalate (Na2C2O4) in acidic solution is titrated with a solution of potassium permanganate (KMnO4) according to the following balanced chemical equation: 2 KMnO4 (aq) + 8 H2SO4 (