Proposed transformation pathways of BPA in the Mn2O3/PMS system. First,... | Download Scientific Diagram
Density functional theory calculated reaction pathways of (a) Mn2O3 +... | Download Scientific Diagram

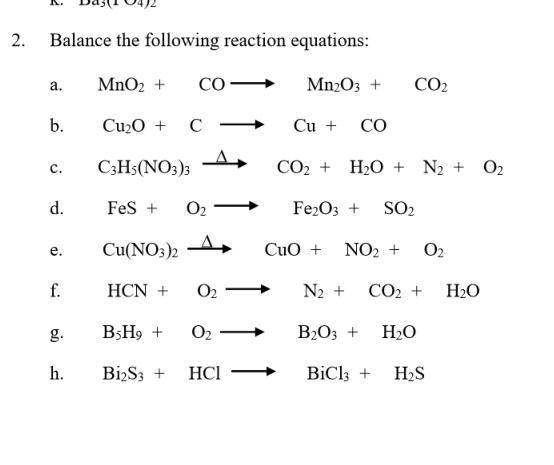
Understanding hydrothermal transformation from Mn2O3 particles to Na0.55Mn2O4·1.5H2O nanosheets, nanobelts, and single crystalline ultra-long Na4Mn9O18 nanowires – topic of research paper in Nano-technology. Download scholarly article PDF and read for ...

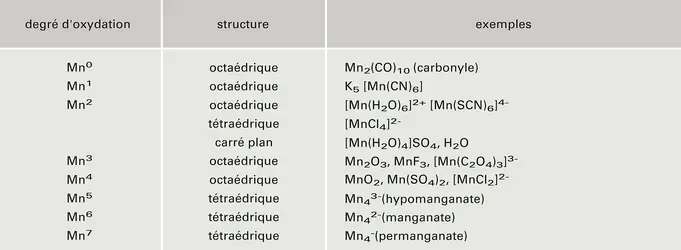
Evaluation of MnOx, Mn2O3, and Mn3O4 Electrodeposited Films for the Oxygen Evolution Reaction of Water | The Journal of Physical Chemistry C
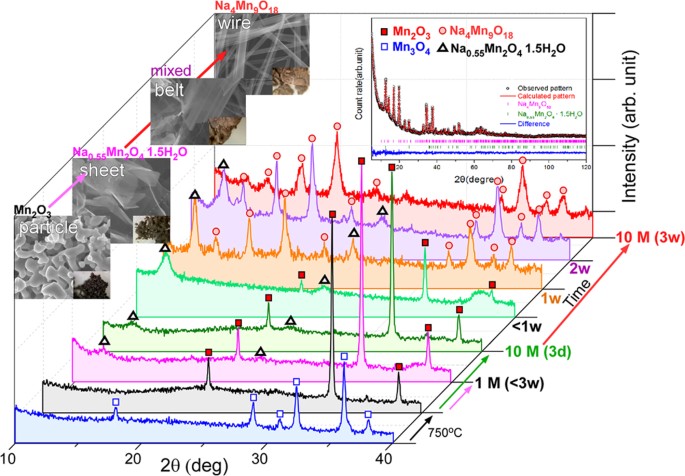
Understanding hydrothermal transformation from Mn2O3 particles to Na0.55Mn2O4·1.5H2O nanosheets, nanobelts and single crystalline ultra-long Na4Mn9O18 nanowires | Scientific Reports

XANES spectra of Mn K-edge; (a) Mn2O3, (b) Mn3O4, (c) MnO2, (d) MnO,... | Download Scientific Diagram

How many grams Mn2O3 would be produced from the complete reaction of 46.8 g of MnO2 ? Zn + 2MnO2 + H2O - brainly.com

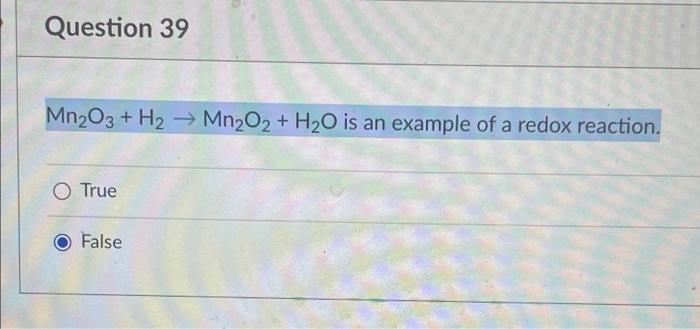
SOLVED: Describe the following reaction in a battery: 2 MnO2(s) + H2O(l) + 2e- —> Mn2O3(s) + 2 H2O(aq) a. MnO2 is oxidized at the anode. b. MnO2 is reduced at the

Irregularly Shaped Mn2O3 Nanostructures with High Surface Area for Water Oxidation | ACS Applied Nano Materials
How many grams Mn2O3 would be produced from the complete reaction of 46.8 g of MnC 2 ? Zn+ [algebra]
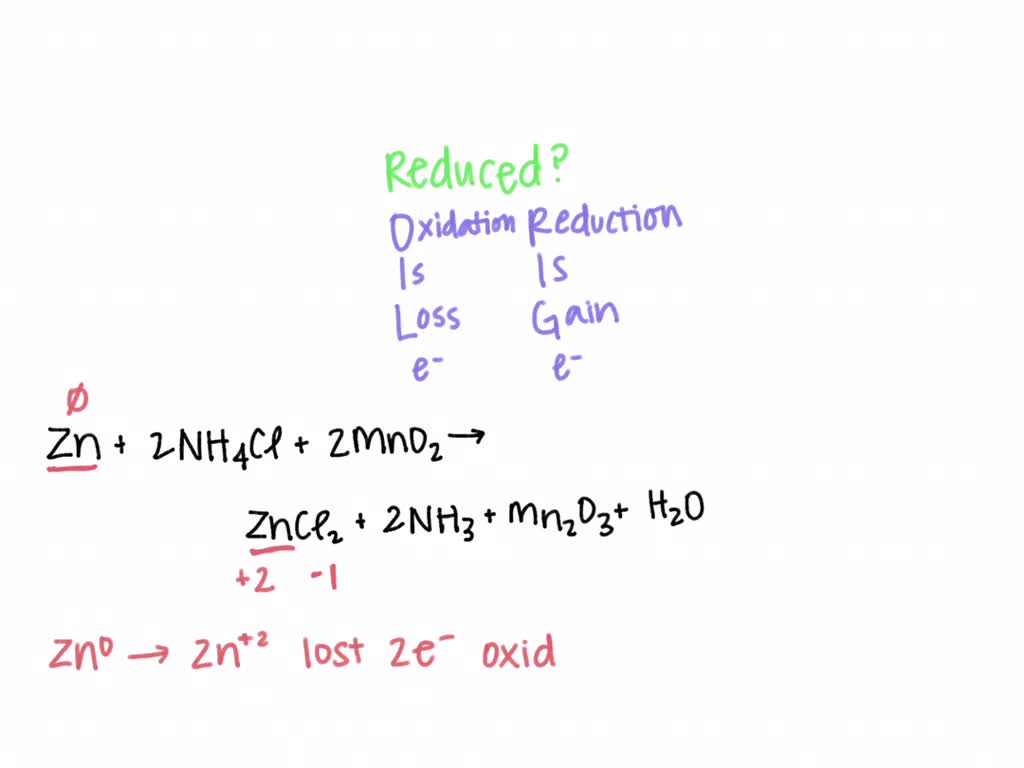
SOLVED: In an acid dry cell battery for which the reaction is Zn + 2 NH4Cl + 2 MnO2 ? ZnCl2 + 2 NH3 + Mn2O3 + H2O, what atom is reduced?




![Solved [3] Manganese dry cell Cathode 2MnO2 + 2H+ + 2e → | Chegg.com Solved [3] Manganese dry cell Cathode 2MnO2 + 2H+ + 2e → | Chegg.com](https://media.cheggcdn.com/study/a0a/a0a0ef03-1105-49d9-a45f-969fa87d8d7f/image)



![Punjabi] Mn2O3 is acidic/basic oxide. Punjabi] Mn2O3 is acidic/basic oxide.](https://static.doubtnut.com/ss/web-overlay-thumb/13363282.webp)