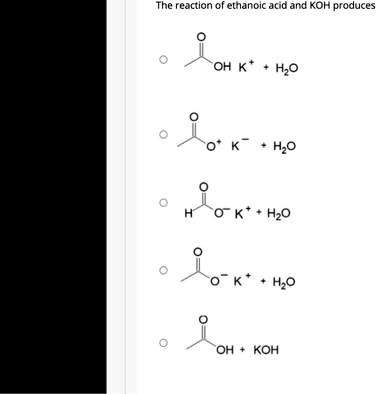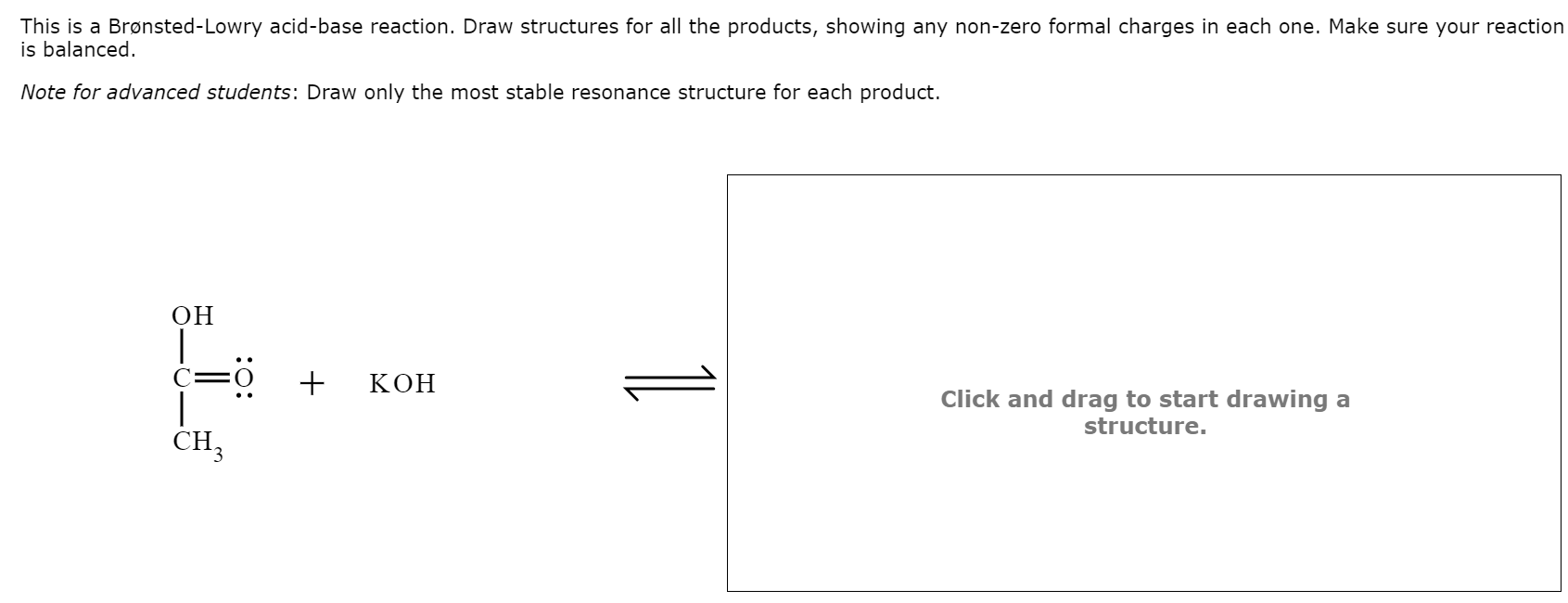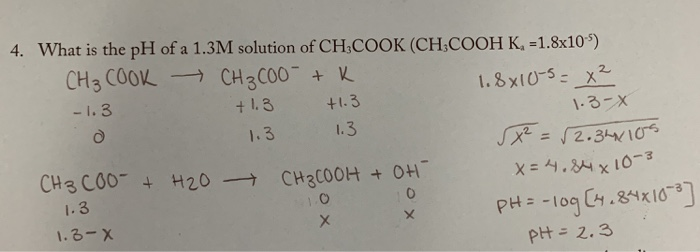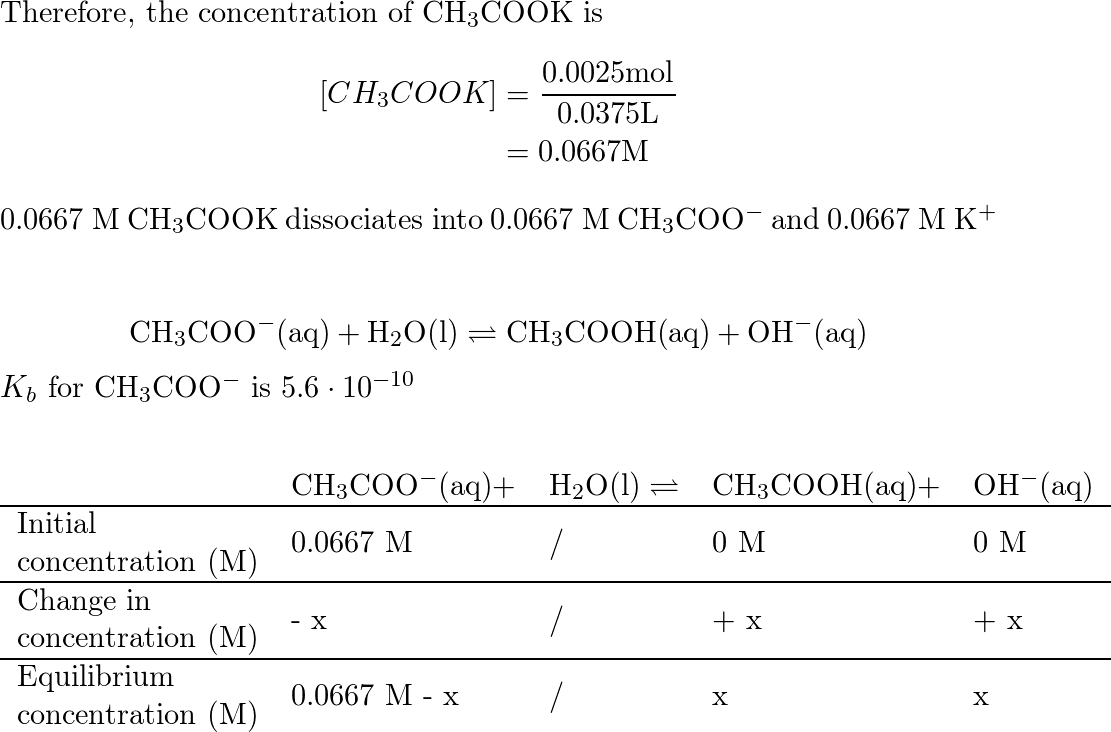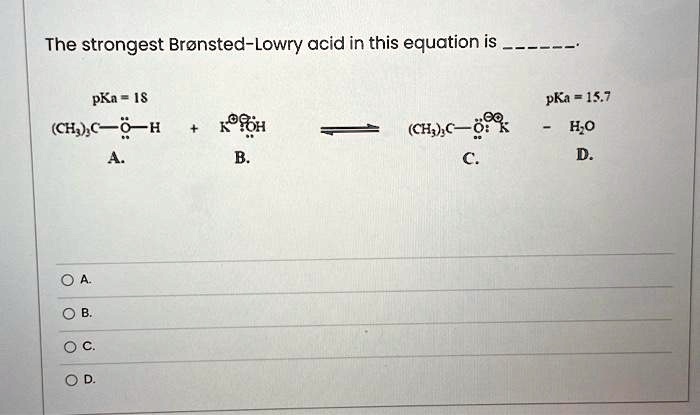
SOLVED: Texts: The strongest Brønsted-Lowry acid in this equation is pKa = 1S pKa = 15.7 - HOD (CH3COOH + KOH → CH3COOK + H2O) (CH3COOH + KOH → CH3COOK + H2O) OA. OB. OC. OD.
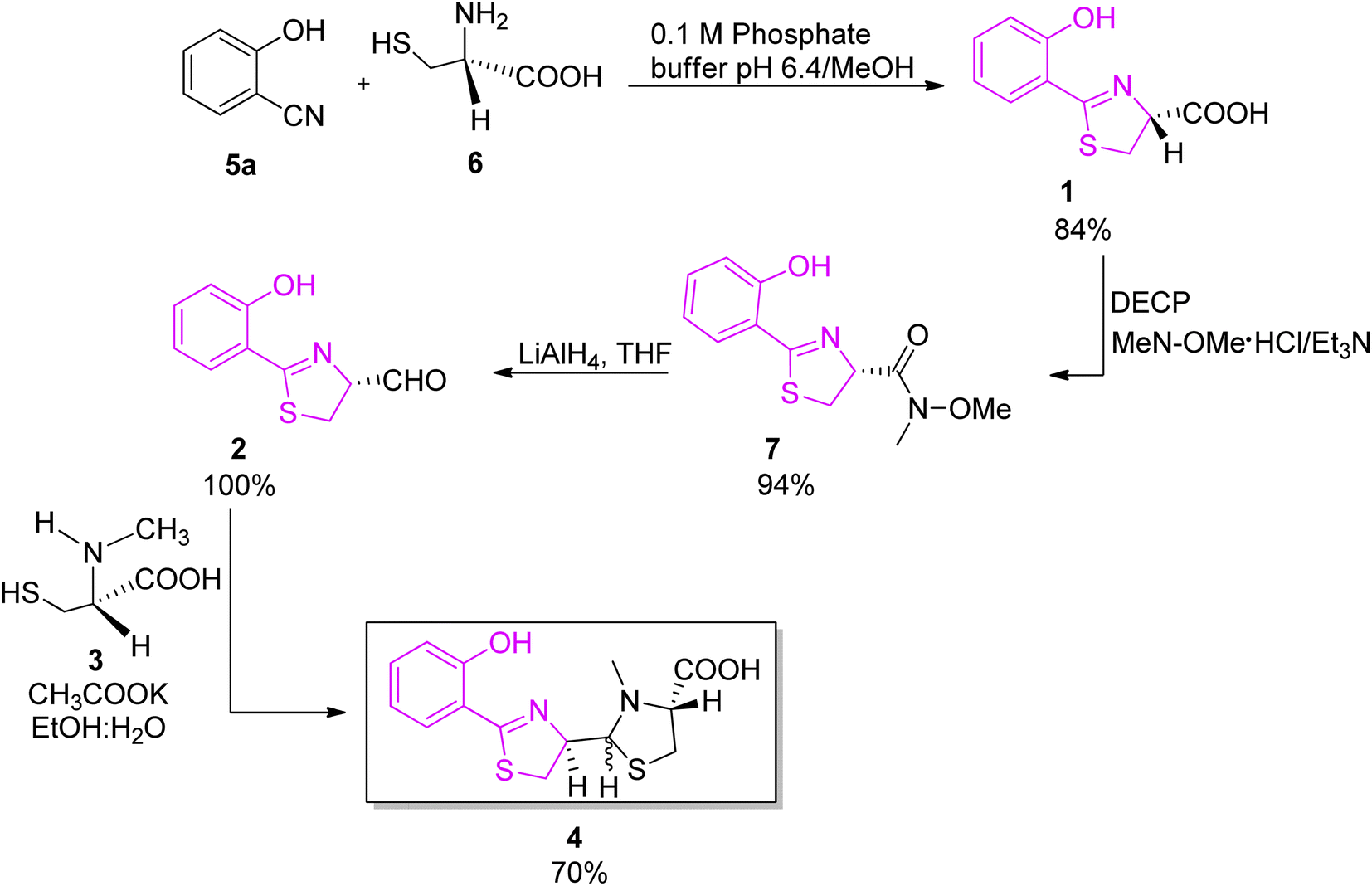
The diversity and utility of arylthiazoline and aryloxazoline siderophores: challenges of total synthesis - RSC Advances (RSC Publishing) DOI:10.1039/D2RA03841B

Preparation of 3β-hydroxy-6-ethylchenocholane derivatives (subset B).:... | Download Scientific Diagram

SOLVED: QUESTION 4 POINT Identify the correct chemical reaction for the neutralization of ethanoic acid by potassium hydroxide. Select the correct answer below: CH3COOH + KOH â†' CH3COOK + H2O CH3COOH +

b) Complete the following reactions i) C6H6 + CH3COCI Anhydrous AICI3 > ii) CH3CH2CH2CH,Br + Na dry ether, iii) CH3COOK +H2O electrolysis →
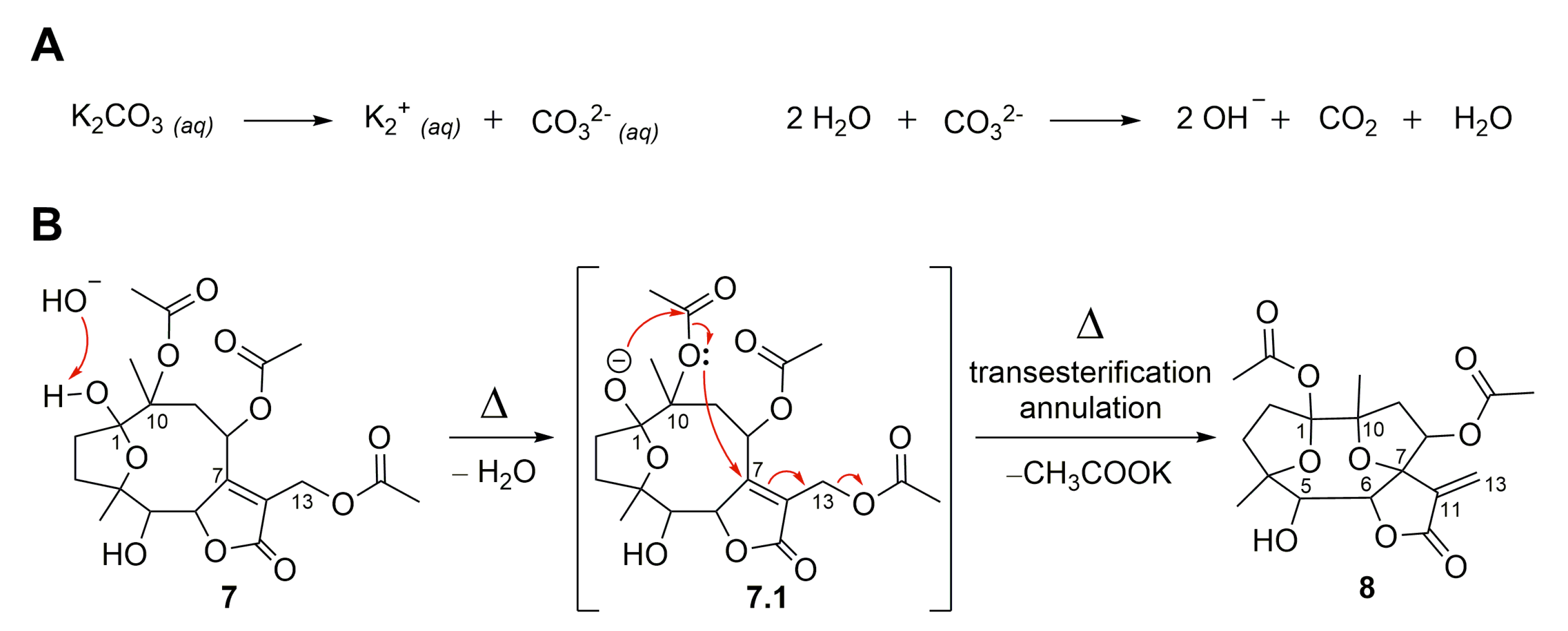
Molecules | Free Full-Text | Semisynthetic Sesquiterpene Lactones Generated by the Sensibility of Glaucolide B to Lewis and Brønsted–Lowry Acids and Bases: Cytotoxicity and Anti-Inflammatory Activities